Abstract
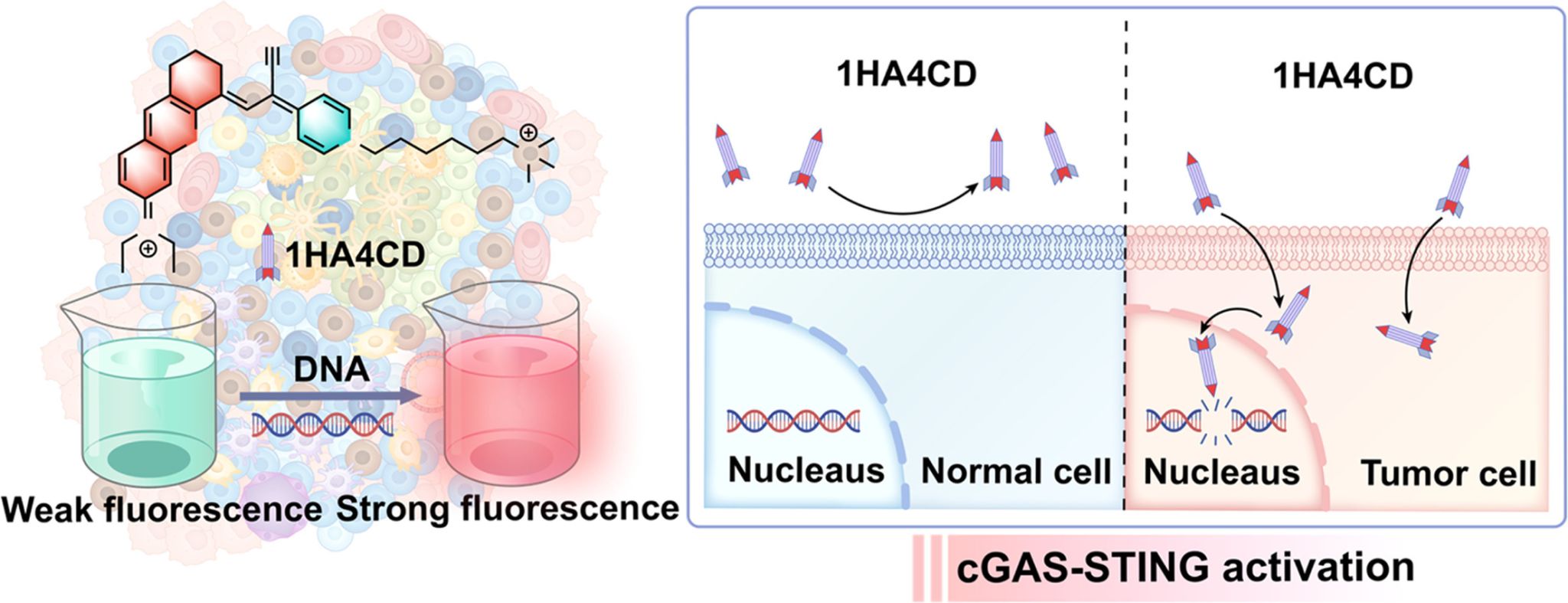
The escalating challenge of chemoresistance in breast cancer treatment severely limits clinical efficacy, necessitating the urgent development of innovative strategies that synergistically enhance tumor cell eradication and remodel the anti-tumor immune microenvironment. To address this, we developed a D-A structured theranostic probe, 1HA4CD, featuring a dihydroxanthene-fluorophore with diethylamino donor and acrylonitrile/pyridyl acceptors. Upon laser irradiation, 1HA4CD enables spatiotemporally controlled reactive oxygen species (ROS, primarily singlet oxygen, 1O₂) generation. Crucially, its precise nuclear localization facilitates the induction of high-concentration ROS within the nucleus, causing irreversible oxidative genomic DNA damage. RNA sequencing analysis revealed that the transient nuclear ROS overload not only directly induces DNA double-strand breaks (DSBs) but also inhibits DNA repair pathways, creating a "dual-hit" effect that effectively overcomes the chemoresistance associated with traditional DNA-damaging agents through a nuclear-targeted photodynamic mechanism. DNA fragments released into the cytoplasm post-damage are recognized by the cytosolic DNA sensing machinery, subsequently activating the cGAS-STING signaling cascade, which leads to the systemic activation of both innate and adaptive immune responses. In vivo animal studies demonstrated that 1HA4CD-mediated photodynamic therapy exhibits significant therapeutic efficacy against breast cancer, coupled with a favorable biosafety profile. This research presents a nuclear-targeted molecular tool for photodynamic immune activation therapy and advances the development of combination therapies based on DNA damage-induced immune responses.
文章链接:https://doi.org/10.1016/j.actbio.2026.01.054