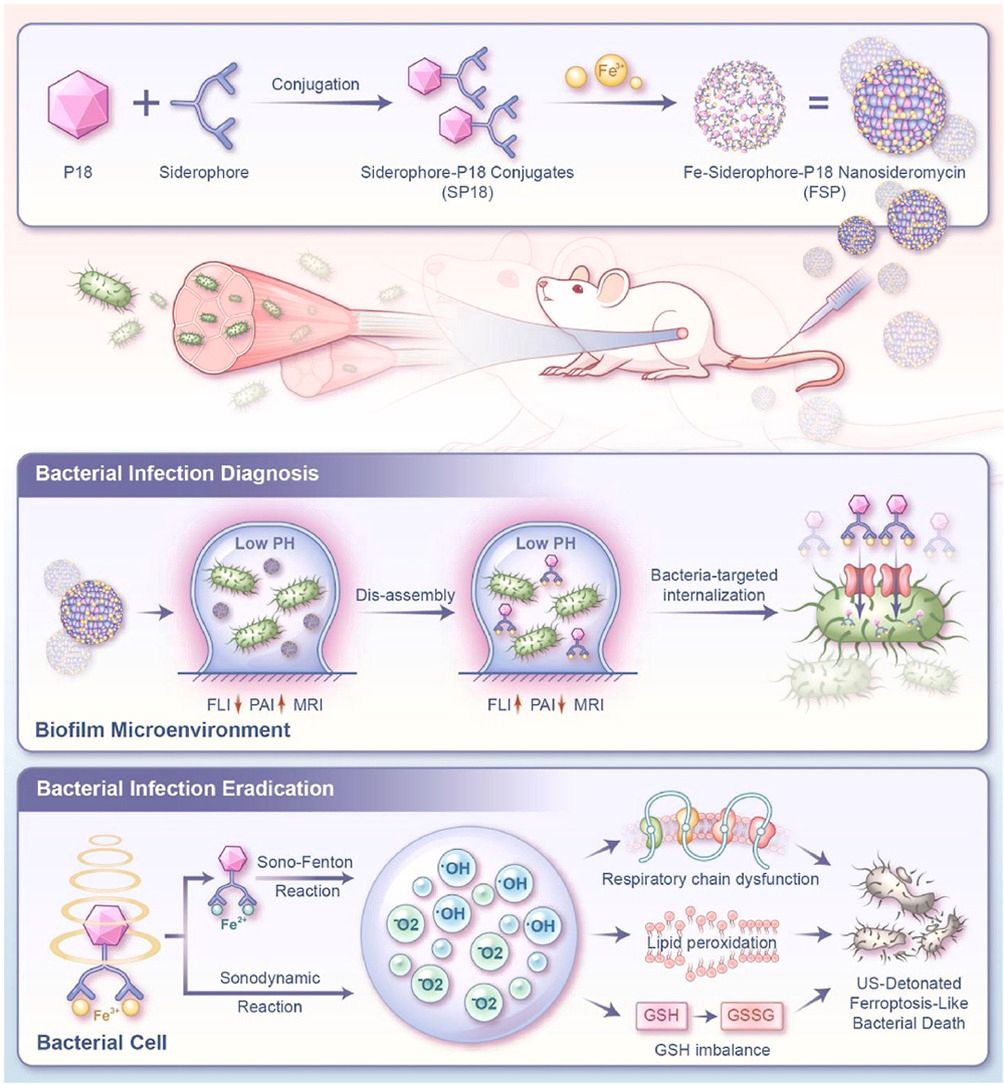
Abstract
Biofilm formation poses a severe challenge to antibacterial stewardship. While siderophore-antibiotic conjugates (termed as sideromycins) offer a promising solution, their efficacy is inherently limited by antibiotic resistance. To transcend this barrier, we pioneer a transformative siderophore-sonosensitizer conjugate through covalent linkage of a catechol siderophore to purpurin 18 (a sonosensitizer). This novel conjugate further self-assembles with iron(III) ions, forming the first-reported carrier-free nanosideromycin—an all-in-on iron-siderophore-sonosensitizer nanoplatform. This design enables ultrasound-denotated reactive oxygen species (ROS) generation and ferroptosis-like amplication. Capitalizing on bacteria-specific siderophore uptake and pH-responsive assembly/disassembly, the nanosideromycin enables precision delivery and active internalization of sonosensitizers into bacteria. This strategy permits real-time localization of infections via concurrent fluorescence/photoacoustic and magnetic resonance imaging. Upon ultrasound irradiation, dual antimicrobial mechanisms of sonosensitizer-mediated sonodynamic therapy and siderophore/iron-augmented sono-Fenton catalysis are stimuonously unleashed, synergistically tirggering an explosive ROS burst and potent ferroptosis-like bacterial death. As a result, mice with multidrug-resistant biofilm-induced pyomyositis were completely cured. Collectively, this first-in-class theranostic nanosideromycin integrates highly-targeted imaging diagnostics, cost-effective yet ultra-efficient ROS generation, and ferroptosis-like bacterial killing, establishing a paradigm-shifting strategy for biofilm therapy with spatiotemporal controllability.
文章链接:https://doi.org/10.1016/j.bioactmat.2025.09.020