Abstract
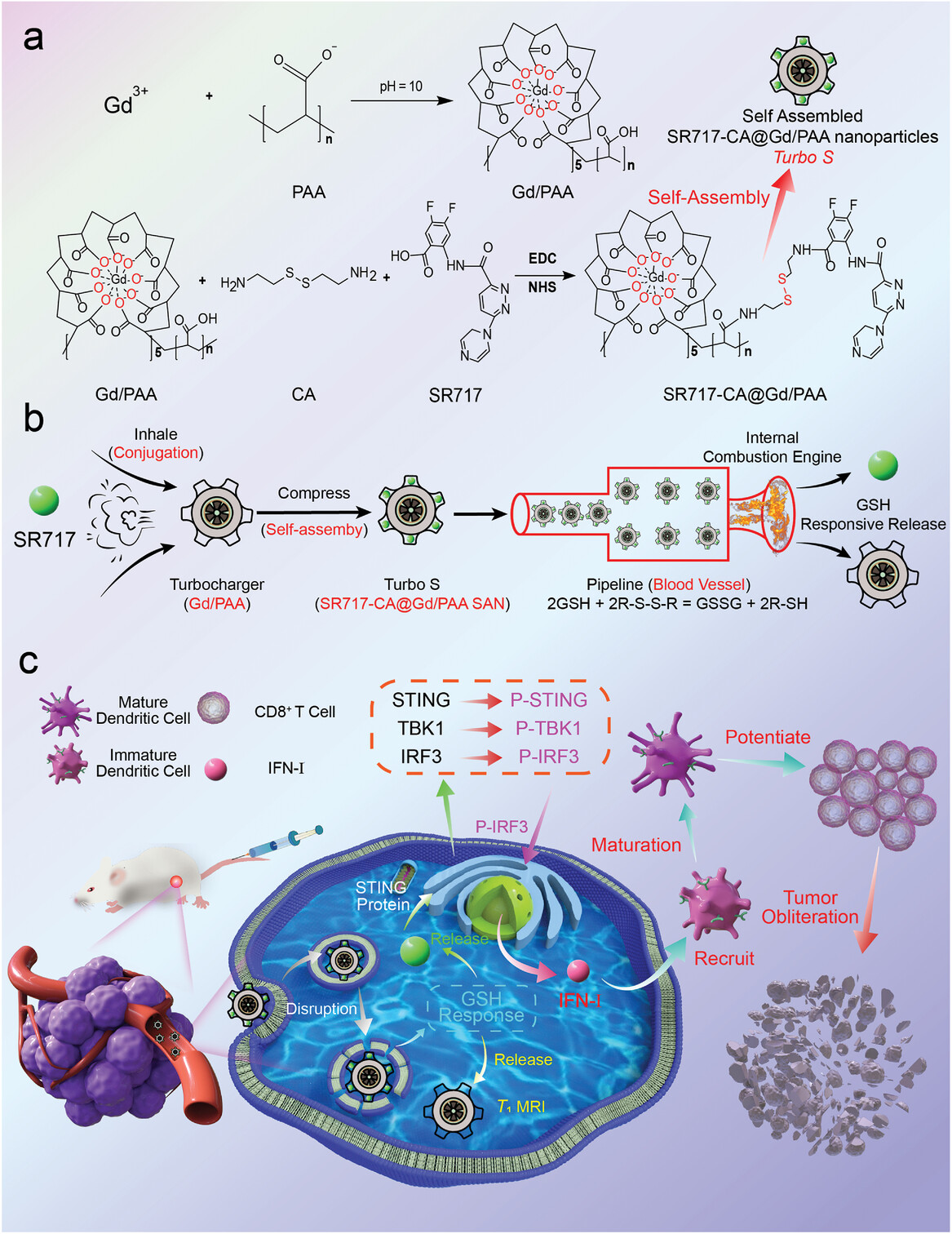
To overcome the problems of Gd-based contrast agents (GBCAs) (nephrotoxicity and brain deposition) and stimulator of interferon genes (STING) agonists (poor stability, low delivery efficiency, and potential toxicity), in this study, a Turbo-charging system-like GBCA is designed and constructed for magnetic resonance imaging (MRI) guided STING pathway-activated cancer immunotherapy. Poly(acrylic acid) (PAA) is used to coordinate with Gd3+, forming a Gd/PAA macrochelate. Both Gd/PAA macrochelate and SR717 are conjugated to cystamine (CA) to obtain SR717-CA@Gd/PAA self-assembled nanoparticles (SAN), which are termed as Turbo S because of its similarity with the Turbo-charging system of cars. After accumulation in tumors and internalization in tumor cells, the disulfide linkage in Turbo S undergoes a cleavage process catalyzed by glutathione (GSH), leading to the release of Gd/PAA and SR717. The released Gd/PAA gain a high r1 value (17.11 mM−1 s−1 at 7.0 T; 57.81 mM−1 s−1 at 3.0 T), indicating its strong T1 imaging capability. Turbo S with a low dosage of SR717 (8.9 mg kg−1) achieved a higher tumor immunotherapeutic efficacy than free SR717 with a high dosage (30 mg kg−1). The excellent delivery efficiency, high tumor treatment efficacy, and superior biosafety demonstrate that the Turbo S can be used as a promising candidate for tumor immunotherapy.
文章链接:https://onlinelibrary.wiley.com/doi/10.1002/advs.202410432